Should I Lime? Understanding the pH of Your Soil
go.ncsu.edu/readext?678050
en Español / em Português
El inglés es el idioma de control de esta página. En la medida en que haya algún conflicto entre la traducción al inglés y la traducción, el inglés prevalece.
Al hacer clic en el enlace de traducción se activa un servicio de traducción gratuito para convertir la página al español. Al igual que con cualquier traducción por Internet, la conversión no es sensible al contexto y puede que no traduzca el texto en su significado original. NC State Extension no garantiza la exactitud del texto traducido. Por favor, tenga en cuenta que algunas aplicaciones y/o servicios pueden no funcionar como se espera cuando se traducen.
Português
Inglês é o idioma de controle desta página. Na medida que haja algum conflito entre o texto original em Inglês e a tradução, o Inglês prevalece.
Ao clicar no link de tradução, um serviço gratuito de tradução será ativado para converter a página para o Português. Como em qualquer tradução pela internet, a conversão não é sensivel ao contexto e pode não ocorrer a tradução para o significado orginal. O serviço de Extensão da Carolina do Norte (NC State Extension) não garante a exatidão do texto traduzido. Por favor, observe que algumas funções ou serviços podem não funcionar como esperado após a tradução.
English
English is the controlling language of this page. To the extent there is any conflict between the English text and the translation, English controls.
Clicking on the translation link activates a free translation service to convert the page to Spanish. As with any Internet translation, the conversion is not context-sensitive and may not translate the text to its original meaning. NC State Extension does not guarantee the accuracy of the translated text. Please note that some applications and/or services may not function as expected when translated.
Collapse ▲pH
The concept of “pH” sure is a strange one to grasp. On one hand we have probably all tasted the difference between the acidity of a lemon and the more bitter taste of a cucumber, but on the other hand, it’s very difficult to visualize how soil pH is affecting our soil health. Especially when trying to understand how it affects plant nutrients and soil bacteria and fungi. So what really is pH and why is it important when it comes to our soils?
What is pH?
pH is a measure of how acidic or basic something is. pH is measured on a scale of 1-14, with 7 being neutral and anything below that being acidic, and above it being basic.
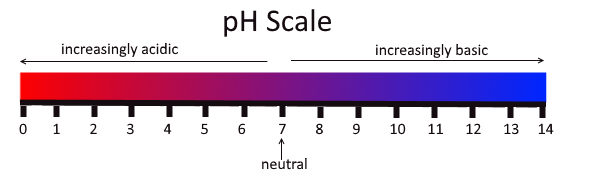
The “p” in pH stands for “power,” and the “H” is the element Hydrogen. This is why the “p” is lowercase and the “H” is uppercase. When we talk about the pH of the soil, or a solution, we are actually talking about the concentration of hydrogen in that solution. Without getting too deep into the chemistry of it all, the more Hydrogen atoms you have, the more acidic the solution. We are talking very minuscule amounts, so in-order to make it easier to comprehend this, it was put on a logarithmic scale to give us the 0-14 pH scale we use today. The biggest thing to take into consideration when comparing two pHs, is that there is a 10x difference between numbers. A pH of 6 is 10x more acidic that a pH of 7!
The biggest thing to take into consideration when comparing two pHs, is that there is a 10x difference between numbers. A pH of 6 is 10x more acidic that a pH of 7!
The middle school science fair vinegar and baking soda volcano is often an example of pH that everyone thinks of when two extremes on the pH scale come into contact with each other. The basic baking soda reacts with the acidic vinegar, there is a violent reaction, and you get the gaseous “volcano” as a result. However our soils are more like the container of stabilized liquid in the bottom of the volcano after the reaction.
Soil pH
Our soils are made of rock pieces that have been around for millions of years slowly changing and degrading with each weather event and seasonal cycle to turn them into soils we rely on today. I like to think of all this degraded rock as “the dirt” and you add in all the fungi, bacteria, organic matter, vertebrates, invertebrates, and other life and this makes it the soil. All of this life intermixed with the rock particles is constantly interacting and shifting with each environmental disruption that occurs. Add into this our farming activities of discing, chiseling, and/or plowing our fields and then fertilizing, planting, watering, and harvesting; and you have a very complex situation for a soil to develop a pH equilibrium.
Why does the amount of hydrogen ions in your soil affect plant growth?
It affects plant growth because different plant nutrients are more/less available at different pHs. It helps to think of the pH’s effect on the soil like a giant bedroom dresser with thousands of drawers. In each drawer there are different nutrients that the plant needs. At different pHs, different sets of drawers are able to be opened and release their nutrients. Soil bacteria and fungi are often the ones opening the drawers and they only work if the pH is the level they like. The nutrients may be in the drawers, but without the right pH, they are locked away in a closed drawer.
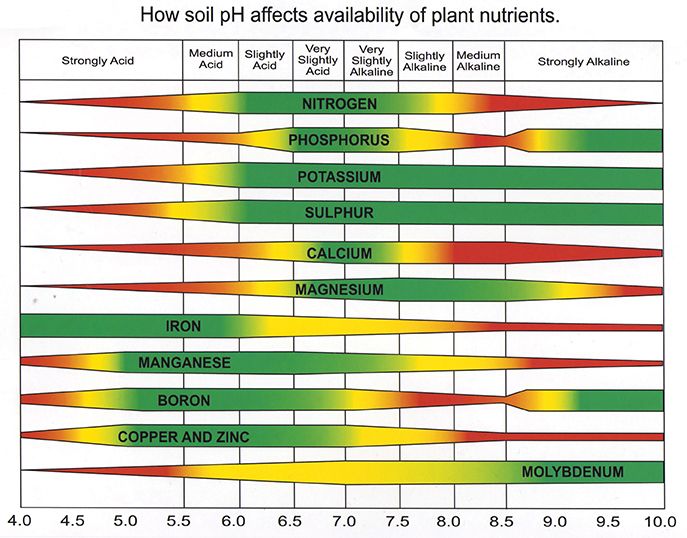 You may have also heard or noticed that different plants prefer different soil pHs. A very common example is that blueberries like acidic soils. Generally when a plant needs a certain pH, this is because that plant evolved in an environment with a specific pH. Just like how humans that evolved in areas without milk consumption do not produce the enzyme required to break down milk sugar, plants that evolve in a specific pH have made physiological compromises to grow in that environment. Blueberries never needed an extremely large root system in an acidic environment because in acidic soil, fungi and acid-loving bacteria flourish and free up nutrients for the blueberries. Move a blueberry into an alkaline soil and it has a hard time getting the nutrients it needs even if those nutrients are right there in the soil. They are locked away in dresser drawers waiting for a pH change.
You may have also heard or noticed that different plants prefer different soil pHs. A very common example is that blueberries like acidic soils. Generally when a plant needs a certain pH, this is because that plant evolved in an environment with a specific pH. Just like how humans that evolved in areas without milk consumption do not produce the enzyme required to break down milk sugar, plants that evolve in a specific pH have made physiological compromises to grow in that environment. Blueberries never needed an extremely large root system in an acidic environment because in acidic soil, fungi and acid-loving bacteria flourish and free up nutrients for the blueberries. Move a blueberry into an alkaline soil and it has a hard time getting the nutrients it needs even if those nutrients are right there in the soil. They are locked away in dresser drawers waiting for a pH change.
So what pH do you need for your growing system and soils? Contact you local Extension agent to get expert help and advice with soil sampling, soil test interpretation, and any pH modification recommendations that may be needed to optimize your production and soil health.
Pasture pH
by Clint Carty
Pastures offer a unique example of soil pH because we are rarely disturbing the soil. So as forage producers and no-till users we have a harder time identifying low pH because it shows up slowly but then becomes a major issue once it onsets. This is why routine soil testing is important for pasture and forage systems. It is good to test your soils once every couple of years to address concerns of pH balance. I recommend soil testing every year though because that gives you a more accurate baseline for fertilizer application and you can start to catch the early onset of pH imbalance. As stated previously by Daniel, the pH affects the availability of nutrients for your forages to uptake and convert into growth.
In the southeast we commonly have soils that are low in pH(acidic) and that is why it is important to lime your pastures and hay fields because they are already naturally more acidic than what is desired for most forage plants. But, it is also important to choose the correct lime for your fields. Calcitic lime contains calcium limestone and dolomitic lime contains calcium and magnesium limestones. Magnesium is an important nutrient for our plants and animals and most farmers in the piedmont region have a magnesium deficiency in their soils which results in the need for a high-mag mineral supplement for their livestock because they are eating forages that are low in the nutrient that their body requires.
pH is one of the biggest factors when it comes to assessing costs associated with a forage system because it is the determining factor of how much nutrients your plants will uptake besides water availability. If your pH is too low then you are faced with spending money on fertilizer when the forages you are trying to improve will only take up a percentage of the nutrients you applied. So why throw your money out the window! Yes, applying lime on a more regular basis will cost more at first but once you can maintain an adequate pH in your soil that’s when it becomes more cost effective because you will be able to spend less money on fertilizer because your plants are taking up a higher percentage of the nutrients you apply. Before you can start crunching numbers and apply soil amendments the first step in a long but rewarding process is……Taking soil samples! The best part is that if you send your samples in the non-peak season, IT’S FREE!!!!!!! So, if you need help give us a call or shoot us an email. Please visit our extension website for contact information.




